Bone metabolism & osteoporosis Diagnostics
Case Study: GeneMedi's GMP-h-β-CTx-Ab pairs (Anti-human β-CTx mouse monoclonal antibody) are validated to detect the GMP-h-β-CTx-Ag01 (Recombinant human β-CTx Protein) in ELISA
Evidence has shown activated osteoclasts create resorption pits with low pH to dissolve the inorganic matrix and lysomal enzymes, such as TRAP and cathepsin K, effectively digest the exposed type-1 collagen releasing specific degradation products, such as the carboxyl-terminal cross-linked telopeptides of type I collagen (CTX-1). The biomarker β-CTx offers significant information on bone metabolism (bone remodeling) and is useful to predict fracture risk and to monitor the treatment efficacy. Recently, our R&D; department demonstrated that our GMP-h-β-CTx-Ab pairs have a large linear range and good sensitivity against the GMP-h-β-CTx-Ag01. Below is the result of GeneMedi's GMP-h-β-CTx-Ab01/02/03/04 (Anti-human β-CTx mouse monoclonal antibody) validation with GMP-h-β-CTx-Ag01 (Recombinant human β-CTx Protein) in ELISA. We highly recommend the Ab01-Ab02 pair and Ag to you.
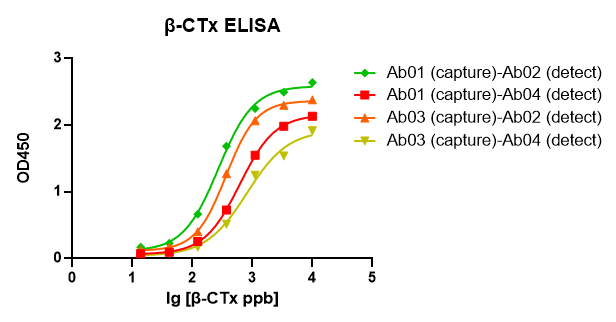
Fig 1. GeneMedi's GMP-h-β-CTx-Ab01/02/03/04 (Anti-human β-CTx mouse monoclonal antibody) are validated to detect the GMP-h-β-CTx-Ag01 (Recombinant human β-CTx Protein) in ELISA.
Case Study: GeneMedi's anti-25-OH-VD3/2 antibody validation with 25-OH-VD3/2 in CLIA and Enzymatic CLIA platform
Complete information about the case study: https://www.genemedi.net/i/diagnostics-chemicals-vitamin-d
Direct chemiluminescence platform
Total 25-OH-VD identification ability
Genemedi's 25-OH-VD sandwich antibodies (GMP-SMT-25-OH-VD3/2-Ab01 and GMP-SMT-25-OH-VD3/2-Ab02) were validated on the direct chemiluminescence platform. The total amount of 25-OH-VD in the sample may be successfully detected thanks to the good cross-reactivity of 25-OH-VD2 and 25-OH-VD3.

Figure 1. Total 25-OH-VD recognition ability; As can be seen from the figure above, 25-OH-VD2 and 25-OH-VD3 exhibit good cross-reactivity.
Sample coincidence rate
The VD sandwich antibody made by Genemedi was validated using a direct chemiluminescence platform
with the industry's gold standard LC-MS/MS as a control to increase the accuracy of the
25-OH-VD immunoassay.
When compared to the mass spectrometry detection results, these antibodies exhibited excellent
consistency, with an R2 of up to 0.9737 and a customer-verified R2 of 0.9538. The sandwich
approach greatly improved immunodetection accuracy.

Figure 2. Self-assessment clinical comparative analysis and Clinical comparative analysis of customer testing; The antibodies showed excellent agreement compared to mass spectrometry detection results, with an R2 as high as 0.9737 and a customer-validated R2 of 0.9538. The sandwich approach greatly improves the accuracy of immunoassays.
Enzymatic chemiluminescence platform
standard curve line
The detection concentration range for Genemedi's 25-OH-VD sandwich antibodies (GMP-SMT-25-OH-VD3/2-Ab01 and GMP-SMT-25-OH-VD3/2-Ab02) is 0.1 to 80ng/mL.

Figure 4. Standard curve; As can be seen from the figure above, Genemedi's 25-OH-VD sandwich antibody has a detection concentration range of 0.1 to 80ng/mL.
Sample compliance rate
Detection employing 25-OH-VD sandwich antibodies (GMP-SMT-25-OH-VD3/2-Ab01 and GMP-SMT-25-OH-VD3/2-Ab02) validated by an enzymatic chemiluminescence platform using an R2 of 0.9760, the data demonstrated strong consistency using mass spectrometry (LC-MS/MS).

Figure 5. Clinical comparative analysis; As can be seen from the figure above, Detection employing 25-OH-VD sandwich antibodies validated by an enzymatic chemiluminescence platform using an R2 of 0.9760, the data demonstrated strong consistency using mass spectrometry (LC-MS/MS).
Case Study: Enhanced Detection of 1,25-OH-VD3 Using GeneMedi's Double-Antibody Sandwich ELISA
The measurement of 1,25-dihydroxyvitamin D3 (1,25-OH-VD3), also known as calcitriol, is critical for evaluating the body's vitamin D status. As the hormonally active form of vitamin D, 1,25-OH-VD3 is essential for the regulation of calcium and phosphate metabolism, impacting bone health and immune function. Assessing its levels is particularly important in diagnosing and managing conditions such as osteoporosis, vitamin D deficiency, and parathyroid disorders.
GeneMedi's products offer a significant advantage in detecting 1,25-OH-VD3 through the use of a double-antibody sandwich ELISA method. This technique is superior to competitive assays for Higher Sensitivity and Specificity, Improved Accuracy, and Ease of Use.
1. Modified sandwich ELISA to detect 1,25-OH-VD3
Experimental procedure:
Coating – anti-1,25-OH-VD3 antibody (GMP-SMT-1,25-OH-VD3-Ab01) 1ug/ml, incubate overnight at 4℃;
Blocking – 3% BSA, incubated 1h at 26℃ (RT);
1,25-OH-VD3 / Recombinant 1,25-OH-VD3 sensor protein (GMP-SMT-Sensor-5) preparation,mix 50ul of 1,25-OH-VD3 sensor protein with the 50 ul of 3-fold diluted 1,25-OH-VD3, The initial concentration of 1,25-OH-VD3 -100ng, incubate 1h at 26℃ (RT);
1,25-OH-VD3 / Recombinant 1,25-OH-VD3 sensor protein (GMP-SMT-Sensor-5) complex binding with anti-1,25-OH-VD3 antibody (GMP-SMT-1,25-OH-VD3-Ab01), Add 100ul of the 1,25-OH-VD3/ Recombinant 1,25-OH-VD3 sensor protein prepared above into the ELISA plate coated with anti-1,25-OH-VD3 antibody, incubate 1h at 26℃ (RT);
secondary antibody, 100 ul of Antiflag HRP (1:5000), incubate 1h at 26℃ (RT).
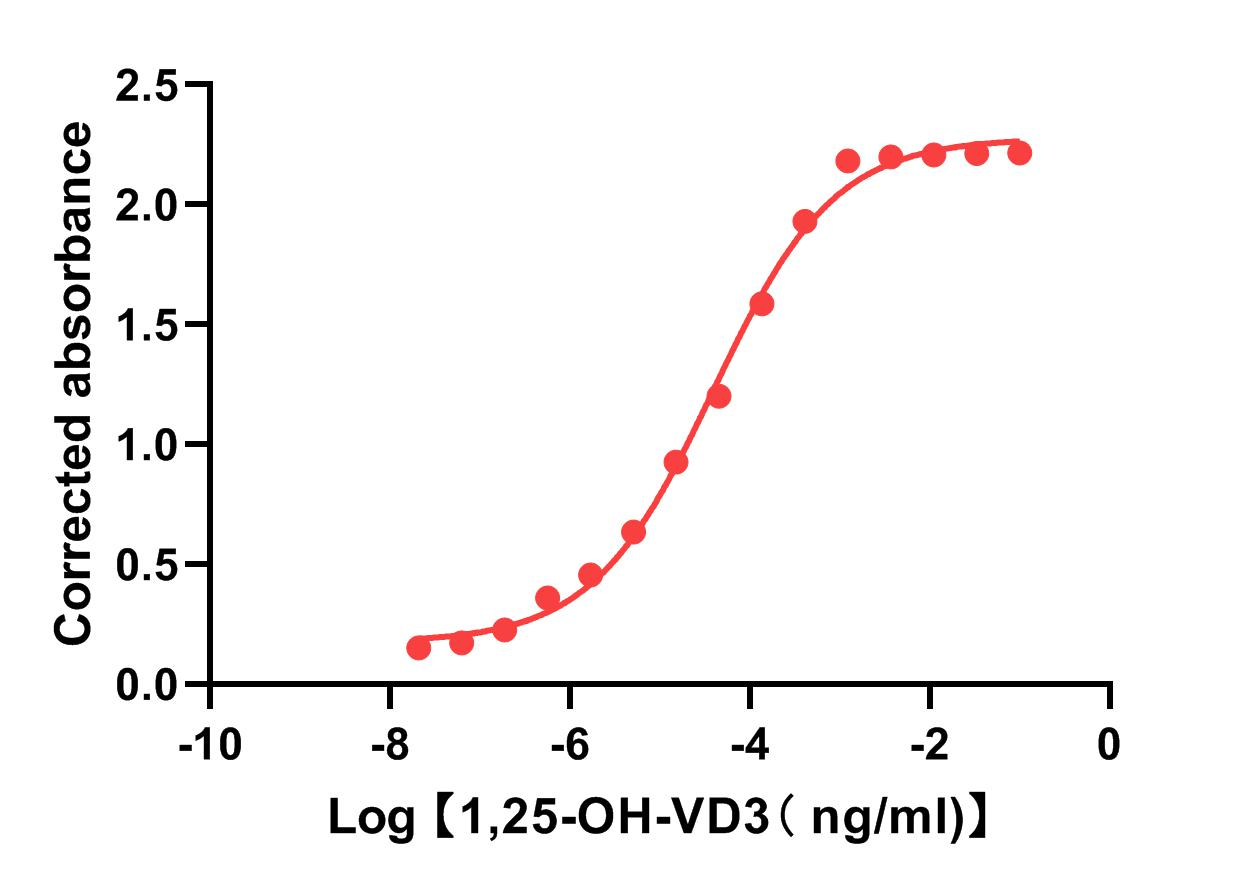
Result:
This method is highly sensitive to 1,25-OH-VD3 detection;
The Ec 50 of the 1,25-OH-VD3 detection using this modified method is 39pg;
The lower detection limit of detecting 1,25-OH-VD3 using this technique is about 1pg.
Figure 1. Modified sandwich ELISA to detect 1,25-OH-VD3. This method is highly sensitive for 1,25-OH-VD3 detection. The Ec 50 of the 1,25-OH-VD3 detection using this modified method is 39pg. The lower detection limit of detecting 1,25-OH-VD3 using this technique is about 1pg.
2.Modified sandwich ELISA – cross-reactivity checking<
Table 1: Products and their forms used in the experiment
| Cat. No | Name | The product is used in experiments | Format |
| GMP-SMT-1-25-OH-VD3-Ag01 | BSA-1,25-hydroxy (OH) Vitamin 3 (1,25-OH-VD3) | NO | Antigen |
| GMP-SMT-1-25-OH-VD3-Ag02 | OVA-1,25-hydroxy (OH) Vitamin 3 (1,25-OH-VD3) | NO | Antigen |
| GMP-SMT-1-25-OH-VD3-Ab01 | Anti-human 1,25-hydroxy (OH) Vitamin 3 (1,25-OH-VD3) mouse monoclonal antibody (mAb) | YES | Antibody |
| GMP-SMT-1-25-OH-VD3-Ab02 | Anti-human 1,25-hydroxy (OH) Vitamin 3 (1,25-OH-VD3) human monoclonal antibody (mAb) | NO | Antibody |
| GMP-SMT-Sensor-5 | Recombinant 1,25-OH-VD3 sensor protein | YES | Protein |
| / | 25-OH-VD3 | YES | Chemical |
| / | 1,25-OH-VD3 | YES | Chemical |
| / | 25-OH-VD2 | YES | Chemical |
| / | Vitamin D3 | YES | Chemical |
| / | Vitamin D2 | YES | Chemical |
Coating: anti-1,25-OH-VD3 antibody (GMP-SMT-1,25-OH-VD3-Ab01) 1 ug/ml;
Blocking: 3% BSA;
In vitro Reactions (Introduction in table 1)
Analyte:
1. 25-OH-VD3 (5 ug/ml)
2. 1,25-OH-VD3 (5 ug/ml)
3. 25-OH-VD2 (5 ug/ml)
4. vitamin D3 (5 ug/ml)
5. vitamin D2 (5 ug/ml)
Fold of dilution – Dilute 3-fold and perform 15 dilutions.
Recombinant 1,25-OH-VD3 sensor protein (GMP-SMT-Sensor-5) – 0.5 ug/ml;
Incubation: Mix 100 ul of diluted vitamin and 100 ul of Recombinant 1,25-OH-VD3 sensor protein (GMP-SMT-Sensor-5) incubate 1 h;
Capture: 1,25-OH-VD3 / Recombinant 1,25-OH-VD3 sensor protein (GMP-SMT-Sensor-5) complex – 100 ul of the invitro reacted samples;
Detection: 100 ul of Anti-flag HRP (1:5000), Incubation temperature 26℃ (RT)/1h
GeneMedi's 1,25-OH-VD3 antibody-1,25-OH-VD3 sensor protein panel demonstrate excellent sensitivity and specificity
GeneMedi's GMP-SMT-1-25-OH-VD3-Ab01 and GMP-SMT-Sensor-5 panel combination demonstrates excellent sensitivity and specificity in the modified sandwich ELISA. In a modified sandwich ELISA assay, the combination of GMP-SMT-1-25-OH-VD3-Ab01 with other analytes, including 25-OH-VD3, 1,25-OH-VD3, 25-OH-VD2, Vitamin D3, and a Vitamin panel, exhibits significantly lower affinity and specificity compared to the combination of GMP-SMT-1-25-OH-VD3-Ab01 and the GMP-SMT-Sensor-5 panel. The latter demonstrates higher affinity and superior specificity in the assay, making it an ideal choice for optimizing biomolecular detection.
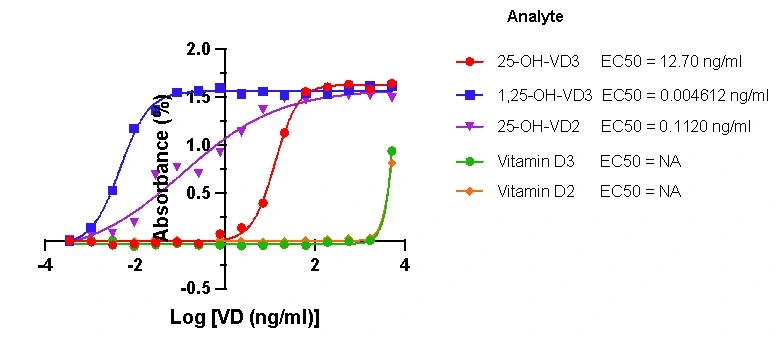
Figure 2. Modified sandwich ELISA – cross-reactivity checking.
This method is highly sensitive for 1,25-OH-VD3. The Recombinant 1,25-OH-VD3 sensor protein has a 200-fold lower affinity with 25-OH VD3 compared to 1,25-OH-VD3. In the modified sandwich ELISA assays, the complex of GeneMedi's 1,25-OH-VD3 antibody with the 1,25-OH-VD3 sensor protein can detect the 1,25-OH-VD3 antigen but is unable to detect other vitamin D proteins (such as 25-OH-VD3, 25-OH-VD2, vitamin D3, and vitamin D2). Therefore, 1,25-OH-VD3 demonstrates higher specificity.
GDU-products for Bone metabolism monitoring& osteoporosis Diagnostics
Novel biological markers have been developed, and studies suggest that they may be valuable research tools for investigating the mechanisms of bone metabolism and assessing the activity of osteocytes, and some of them may be of value for the management of patients with osteoporosis and other bone diseases. To better predict fracture risk and monitor the treatment efficacy of osteoporosis and other bone diseases, GDU offers diagnostic antibodies and antigens for the biomarkers, such as CTX-1, P1NP, N-MID OC, PTH and 25-OH-VD. These biomarkers can be used on the bone metabolism panel.
| Cat No. | Biomarker | Physiological process | Clinical value |
| GMP-h-β-CTx | β-isomerized C-terminal telopeptides (β-CTx), Beta-CrossLaps (beta-CTx) | Bone resorption | Evaluate bone resorption rate |
| GMP-h-PINP | procollagen type I N-terminal propeptide (PINP), amino-terminal propeptide of type 1 procollagen (P1NP) |
Bone formation | Evaluate bone formation rate |
| GMP-h-N-MID-OC | N-terminal midfragment of Osteocalcin (N-MID OC) | Bone formation | Evaluate bone formation rate |
| GMP-h-PTH | parathyroid hormone (PTH) | Calcium cycle | Evaluate hypercalcemia/ hypocalcemia/ hyperparathyroidism |
| GMP-SMT-25-OH-VD3/2 | 25 hydroxyvitamin D (25-OH-(VD3+VD2)) | Calcium cycle | Evaluate vitamin D sufficiency |
| GMP-SMT-25-OH-VD-3 | 25-hydroxy (OH) Vitamin D3 (25-OH-VD-3) | Calcium cycle | Evaluate vitamin D sufficiency |
| GMP-SMT-1-25-OH-VD3 | 1,25-hydroxy (OH) Vitamin 3 (1,25-OH-VD3) | Calcium metabolism | Osteoporosis, vitamin D deficiency |
Introduction of Bone metabolism (bone remodeling), calcium cycle, and osteoporosis
Bones are not inert structures within the human body; they continue to change over the course of a
lifespan. Bone metabolism is characterized by an intimate cooperation of bone cells including
osteoblasts, osteoclasts, and osteocytes in order to maintain bone tissue quantity and the integrity
of bone structure. This process of skeletal change is also known as bone remodeling, which both
protects the structural integrity of the skeletal system and metabolically contributes to the body's
balance of calcium and phosphorus. Remodeling entails the resorption of old or damaged bone,
followed by the deposition of new bone material. It is mainly regulated by osteoclastic bone
destruction and osteoblastic bone formation. Besides, osteocytes also have a role in this process as
well.
The bone remodelling cycle lasts 150–200 days and is primarily mediated by osteoblastic signals
which promote the differentiation and maturation of osteoclast precursors. Activated osteoclasts
create resorption pits with low pH to dissolve the inorganic matrix and lysomal enzymes, such as
TRAP and cathepsin K, effectively digest the exposed type-1 collagen releasing specific degradation
products,such as the carboxyl-terminal cross-linked telopeptides of type I collagen (CTX-1).
Osteoblasts are attracted to this eroded surface and begin to form new osteoid. Type-1 collagen,
abundant in osteoblasts, is secreted as a procollagen precursor molecule into the extracellular
space where it is cleaved at the amino- and carboxy-terminals releasing pro-peptides into the blood.
The procollagen type I N-terminal propeptide (P1NP) and osteocalcin (OC) are the the most sensitive
markers of bone formation rate. Although labile, assay of intact osteocalcin (iOC) is established as
the standard assay for evaluating osteoblastic function. Study examines the clinical usefulness of
the newly developed immunoradiometric assay for osteocalcin (OC), which identifies the stable
N-terminal midfragment of OC (N-MID OC assay). The findings suggest that N-MID OC immunoreactivity
is much more stable than iOC immunoreactivity and that N-MID OC assay may be less susceptible to the
OC fragments reported to accumulate in uremic serum. It may, therefore, prove more reliable than iOC
assay for evaluating bone turnover, and thus for reflecting bone loss. Initially hydroxyapatite
crystals are deposited in the osteoid then a slower mineralisation process continues over several
months, followed by a period of quiescence. RANKL, an essential osteoclastogenic cytokine, is
expressed on the surface of osteoblasts, it binds to its cellular receptor RANK on pre-osteoclasts
and promotes their differentiation and activation. OPG a decoy receptor for RANKL, is secreted by
osteoblasts and other stromal derived cells and reduces bone resorption by binding to RANK and
preventing osteoclastic activity.
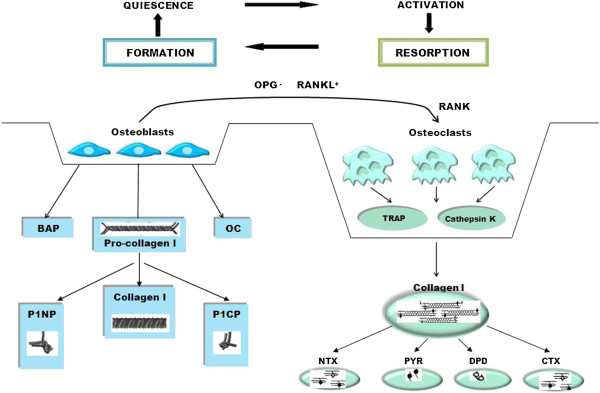
Fig 2. Bone metabolism (bone
remodeling) cycle.
Calcium as a nutrient is most commonly associated with the formation and metabolism of bone. Bone
tissue serves as a reservoir for and source of calcium for these critical metabolic needs through
the process of bone remodeling. Calcium metabolism is regulated in large part by the parathyroid
hormone (PTH)–vitamin D endocrine system, which is characterized by a series of homeostatic feedback
loops. The rapid release of mineral from the bone is essential to maintain adequate levels of
ionized calcium in serum. During vitamin D deficiency states, bone metabolism is significantly
affected as a result of reduced active calcium absorption. This leads to increased PTH secretion as
the calcium sensing receptor in the parathyroid gland senses changes in circulating ionic calcium.
Increased PTH levels induce enzyme activity (1α-hydroxylase) in the kidney, which converts vitamin D
to its active hormonal form, calcitriol. In turn, calcitriol stimulates enhanced calcium absorption
from the gut. Not surprisingly, the interplay between the dynamics of calcium and vitamin D often
complicates the interpretation of data relative to calcium requirements, deficiency states, and
excess intake.
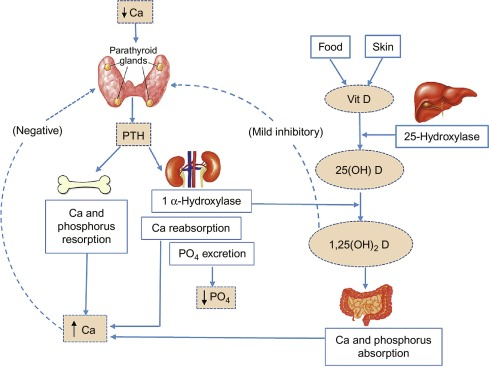
Fig 3. calcium
cycle.
In metabolic bone diseases such as osteoporosis, bone metabolism is altered, leading to bone loss
that is often accompanied by changes in the microarchitecture that result in bone fragility and
ultimately fracture.
Osteoporosis Diagnostics and Therapy Monitoring: Bone remodeling markers and calcium cycle markers
Osteoporosis is a systemic skeletal disorder characterized by low bone mass, micro-architectural
deterioration of bone tissue leading to bone fragility, and consequent increase in fracture risk. It
is the most common reason for a broken bone among the elderly. Bones that commonly break include the
vertebrae in the spine, the bones of the forearm, and the hip. Until a broken bone occurs there are
typically no symptoms. Bones may weaken to such a degree that a break may occur with minor stress or
spontaneously. After the broken bone heals, the person may have chronic pain and a decreased ability
to carry out normal activities. Monitoring of patients treated for osteoporosis should include
regular contact with a health care professional to ensure that: (1) medication is taken regularly
and correctly, (2) calcium and vitamin D intake are sufficient, (3) the patient has no adverse
effects or fear of adverse effects that must be addressed, and (4) there are no comorbidities or
other medications that might alter the expected treatment effect.
The development of serum and urinary assays for biochemical markers, reflecting either enzymatic
activities of osteoblasts and osteoclasts or breakdown products of bone tissue, has been very useful
for investigating the complex pathways of bone metabolism and their alterations in bone diseases.
The biomarkers, such as CTX-1, P1NP, N-MID OC, PTH and 25-OH-VD, offer significant information on
bone metabolism (bone remodeling) and calcium cycle and are useful to predict fracture risk and to
monitor the treatment efficacy.






 Bone metabolism & osteoporosis Products List Download
Bone metabolism & osteoporosis Products List Download